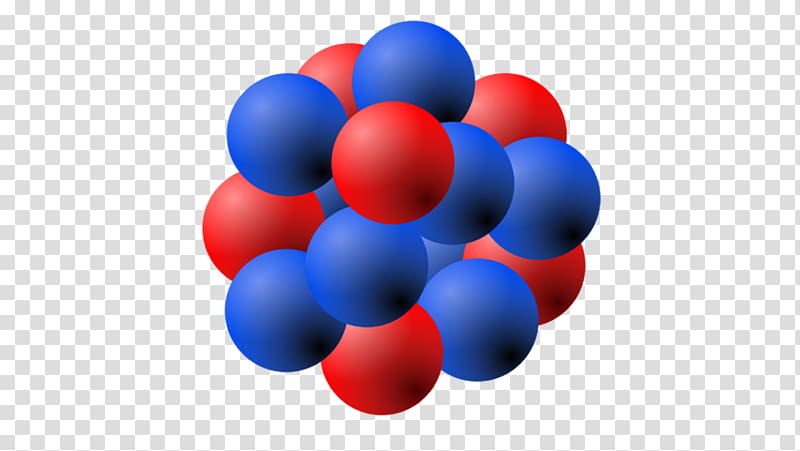
The isotope of an atom is based on the number of neutrons in the nucleus. If the nucleus can be considered to have a radius of the order of 5 fm (= 10 × 10 -15 m), this means its cross-section is of the order of 10 -28 m 2, and its volume is about 10 -42 m 3. The nucleus is more or less a spheroid and may be somewhat prolate (long) or oblate (flat) or otherwise not entirely round. These baryons are further made up of sub-atomic fundamental particles known as quarks joined by the strong interaction. The nucleus of an atom is made up of protons and neutrons (two types of baryons) joined by the nuclear force. Lewis wrote in his famous article The Atom and the Molecule that "the atom is composed of the kernel and an outer atom or shell". The use of the word nucleus in atomic theory, however, did not happen immediately. The modern atomic meaning was proposed by Ernest Rutherford in 1912. In 1844, Michael Faraday used nucleus to describe the “central point of an atom”. The word nucleus is from 1704, meaning “kernel of a nut”. The nucleus is actually held together by another force known as the strong nuclear force. 
Unless there was something else holding the nucleus together it could not exist because the protons would push away from each other. Things that have the same charge repel each other: this repulsion is part of what is called electromagnetic force. Because the nucleus is only made up of protons and neutrons it is positively charged. Neutrons have no charge and protons are positively charged. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus with a very small contribution from the orbiting electrons. Although it is only a very small part of the atom, the nucleus has most of the mass. These sizes are much smaller than the size of the atom itself by a factor of about 23,000 (uranium) to about 145,000 (hydrogen). The size (diameter) of the nucleus is between 1.6 fm (10 −15 m) (for a proton in light hydrogen) to about 15 fm (for the heaviest atoms, such as uranium). It is made up of nucleons called ( protons and neutrons) and is surrounded by the electron cloud. In the nucleus, the protons are in red and neutrons are in purple
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
